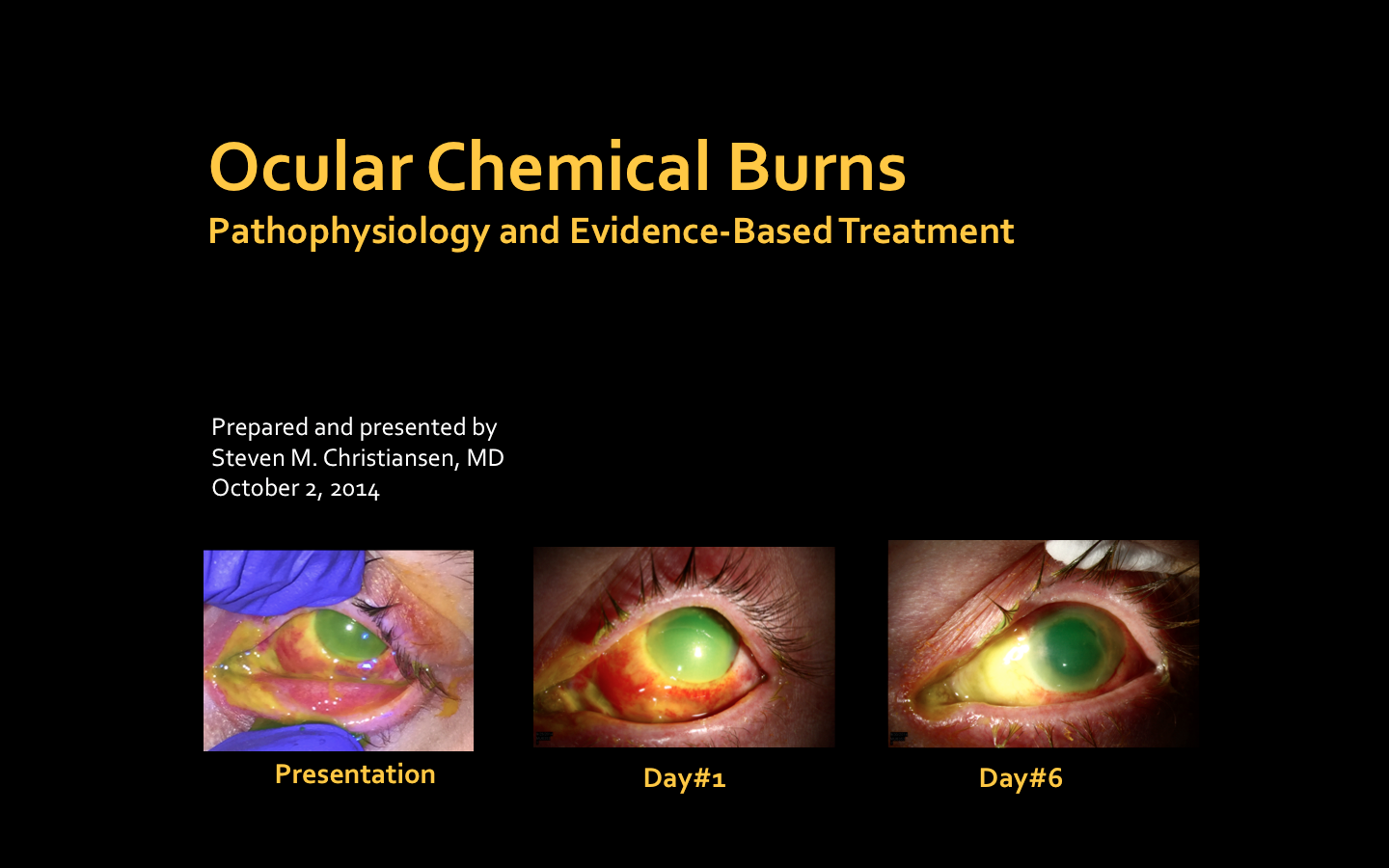
Russian scientists have successfully developed an advanced model of chemical corneal burns in mice. This breakthrough is expected to pave the way for novel human treatment strategies to prevent blindness resulting from such injuries, as announced by the Russian Science Foundation (RSF).
The Challenge of Corneal Burns
Chemical burns to the cornea represent a formidable challenge in ophthalmology. They frequently lead to irreversible tissue damage and vision loss, primarily due to the formation of opaque scars and the ingrowth of blood vessels into the cornea. Current therapeutic approaches, often involving anti-inflammatory or anti-scarring agents, frequently lack sufficient efficacy and can induce significant side effects. In severe cases, a corneal transplant is the sole option, but this procedure sometimes results in graft rejection or clouding.
Improving Experimental Models
Developing effective treatments for these conditions is complicated by the limitations of existing chemical burn models. These models, which involve applying alkali or acid to the eyes of laboratory animals, suffer from a high measurement error, estimated at approximately 40%, due to their experimental methodology.
The MSU Breakthrough
Researchers at Lomonosov Moscow State University refined the chemical corneal burn model in mice. By enhancing the experimental technique, they significantly reduced measurement variability. The team utilized varying concentrations of sodium hydroxide (alkali) to induce distinctly different corneal healing outcomes, hypothesizing that low-concentration burns would lead to wound regeneration, while high concentrations would result in scar formation.
Key Findings on Myofibroblasts
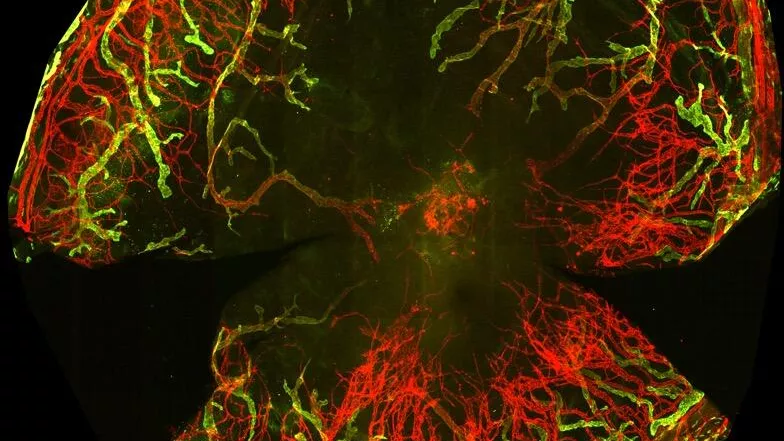
Using this refined model, scientists meticulously tracked cellular and molecular changes within the corneas of laboratory mice. They observed that with a 20 grams per liter alkali concentration, the number of myofibroblasts—cells crucial for wound closure but also implicated in scarring—decreased within seven days post-burn. After three weeks, these cells completely vanished, correlating with a restoration of corneal transparency. In contrast, when a 40 grams per liter alkali concentration was applied, myofibroblasts persisted even after 21 days, including within the endothelium (the inner layer of the cornea), which led to corneal opacity. The higher sodium hydroxide concentration also triggered rapid ingrowth of blood and lymphatic vessels into the deep corneal layers. By the seventh day of the experiment, these vessels were already covered by smooth muscle cells, leading to their stabilization and the subsequent loss of corneal transparency.

Implications for Future Treatment
These findings hold significant importance for both experimental and clinical medicine. They deepen the understanding of the natural mechanisms behind corneal regeneration after injury, facilitate the testing of new anti-scarring drugs, and open new avenues for developing methods to accelerate corneal cell regeneration.
Researcher`s Perspective
“We are confident that our model will become a powerful tool for developing fundamentally new treatments for corneal damage. A crucial discovery was identifying the critical role of myofibroblasts in the healing process. We demonstrated that the speed of their disappearance dictates whether corneal transparency is restored or a scar forms. Moving forward, we plan to use this model to determine the source of myofibroblasts and to uncover which molecular and cellular regulators influence different healing outcomes based on injury severity.”
— Pavel Makarevich, Project Head, Lomonosov Moscow State University